 
Now we need to convert the moles to grams of these elements 0.006430 mol C xįind the mass of Oxygen by subtracting the C and H from the total mass of the sampleĠ.1005g= 0.07716 g C + 0. eXAmple 4.5 Using Combustion analysis to determine an empirical formula of a Compound Containing C, h, and o Problem Suppose you isolate an acid from clover. 
What is the empirical formula for menthol? 0.2829 g of CO 2 x 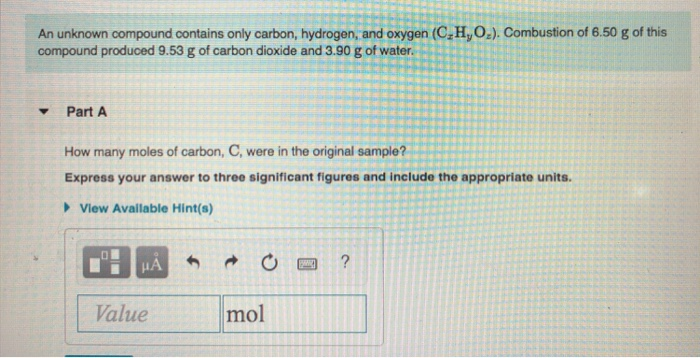
What is the empirical formula of the compound if you. Answer and Explanation: 1 Given, Amount Unknown Compound A 6.60 g Amount of formed Carbon dioxide 15.5 g Amount of formed water 4.24 g The unknown compound A has the elements C. 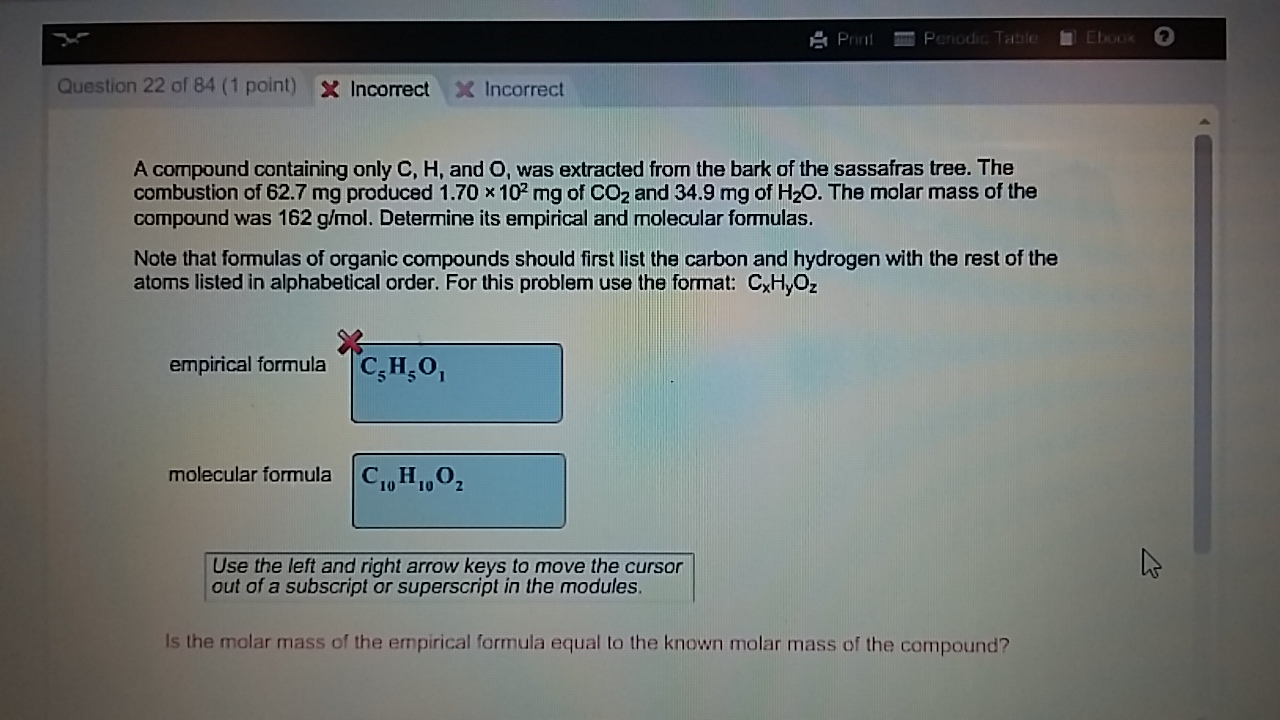
A 0.1005 g sample of menthol is combusted, producing 0.2829 g of CO 2 and 0.1159 g of H 2O. If oxygen is part of the unknown compound, then its oxygen winds up. Menthol, the substance we can smell in mentholated cough drops, is composed of C, H, and O. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
